The taurine controversy
In this article, I explain why I remain cautiously optimistic about taurine as a longevity-promoting supplement despite recent controversies.
Taurine emerges as a key longevity molecule
Let us start with a brief recap of the study that put taurine into the spotlight. In 2023 Parminder Singh published a very intriguing paper under the lead of Vijay Yadav from Columbia University.
The highlights from the paper are:
taurine declines in aging mice, macaque monkeys and humans
taurine extends lifespan of long-lived male and female B6 mice in a well-powered lifespan study
taurine extends the lifespan of worms, but not yeast, suggesting this lifespan-extending effect is conserved in some model species
taurine improves healthspan of aging mice (Fig. 2)
taurine affects various hallmarks of aging and specifically dampens mTOR activity
weak cross-sectional human data linking taurine with health outcomes
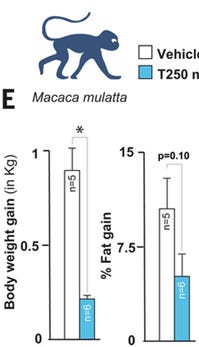
taurine has positive effects on biomarkers in middle-aged macaque monkeys, although we do not know whether this is upstream or downstream of weight-loss
Even before the newly published work criticizing some of these key findings, I had a few reservations. If taurine decreases bodyweight in both mice and monkeys, and mTOR signaling in mice, this points to a CR-mimetic effect. Perhaps taurine has no benefits that are independent of mTOR inhibition, in which case one might just take a low dose of rapamycin which is supported by many more studies.
Excerpt from Figure 4.
Despite these caveats, I really liked the Singh study. It is exceptionally rare for a GRAS supplement to robustly extend mouse lifespan.
Not a biomarker anymore - The downfall of taurine?
Just after the Singh study was published, we heard rumors that others could not reproduce the longitudinal decline in blood taurine seen in mice (claim 1, above).
Confirming this suspicion, in this recent work (Fernandez et al. 2025), a group led by Rafa de Cabo, analyzed data from three human cohorts, mice in the SLAM study and primates. Their data shows that not only does taurine fail to decline with age, in fact, it even increases. No consistent correlations between taurine and markers of health were found across cohorts. The authors speculate that the earlier work by Singh et al. might have captured a spurious correlation because it was based on cross-sectional analysis, which may not be suited for an analyte that shows high inter-individual variability.
I don’t think that is an adequate explanation since Fernandez et al. finds the same changes in cross-sectional data (e.g. PREMED and Balearic cohort).
From what we can tell the absolute levels of taurine measured between the studies were quite similar. In the Singh study the serum taurine levels were below 100 uM for adults and the plasma taurine levels in the Fernandez paper were in a similar range. The Singh study used LC-MS and untargeted metabolomics to determine taurine levels and the Fernandez paper also used a similar technique.
Re-reading the methods section of the Singh paper, I noticed that the main figure contains taurine levels from two or three different sources. Unfortunately, the authors choose not to color-code or discuss this potential shortcoming. It is not clear if blood samples from all these studies were processed in the same way and measured as one batch. Combination of datasets can lead to misleading correlations. Indeed, it appears that most of the decline reported by Singh et al. in humans is attributable to changes between 0-20 years and the data could be compatible with no change of taurine from adulthood to late life, as reported by Fernandez et al.
However, there could certainly still be a benefit to high taurine levels observed in children, entirely consistent with the Singh work. Or high taurine levels could be beneficial even if they don’t decline with age.
Almost at the same time, another group published data from a small cohort confirming that taurine does not decline with age (Marcangeli et al. 2025).
Prior literature on taurine levels and aging
After the Singh paper came out I did a brief read through the literature and came away with the impression that these earlier studies support a decline in (serum) taurine during aging consistent with the new paper. This is a good opportunity to take another look.
Several studies reported serum or blood taurine compared between adult rats and older rats (e.g. Dawson et al. 1996, Pierno et al. 1998). Multiple studies authored by Dawson found a decrease while the latter study by Pierno et al. did not. A major shortcoming of these papers is the small sample size and modest decline reported (1). While there appear to be multiple publications showing an age-related decline, all of them were done in F344 rats by the same lab, limiting generalizability.
It is certainly possible that taurine declines in rats, but not humans or mice, as shown by the Fernandez paper. Alternatively, the above rat studies might not reflect the true changes in rats as a species due to the limited genetic diversity of F344 rats and the small sample sizes used.
My own observations regarding reproducibility
We know from our own work and background research that point (3) - conserved lifespan extension across species - was also not as strong as the authors made it seem. For example, there is no consistent data showing lifespan extension of taurine-fed flies in the literature, which was never mentioned in the Science publication. The effect of taurine on C. elegans lifespan is also less reliable than we hoped. I do not doubt that the authors honestly reported the lifespan extension in C. elegans. However, lifespan extension in worms is often fickle and hard to replicate between labs that use different conditions, especially with compounds that have minor benefits - like 10 to 20% lifespan extension.
Does taurine promote cancer?
A recent study found that bone-like cells in the hematopoietic niche secrete taurine to promote the growth of leukemic cells (Mayerhofer et al. 2025, Sharma et al. 2025). The authors use artificially engrafted leukemic cells and do not study naturally developing leukemia.
Therefore, it is not even clear whether taurine promotes the development of age-related leukemia in mice, let alone humans, or if this effect is large enough to matter. Since B6 mice are quite susceptible to cancer and yet managed to live longer when fed taurine, this implies that any harmful effects are small if they exist at all. It may very well be that T plays a role in a subset of human leukemias which are subset of all cancers which are a subset of all age-related deaths. It would be wonderful if we could develop drugs that target taurine metabolism in these cells, but such a treatment has nothing to do with normative aging.
Overall, I am not particularly worried about this kind of “niche” study. Many beneficial repair, maintenance and cellular defense pathways are implicated in cancer, e.g. Nrf2 signalling or telomerase activation, and yet activation of these generally promotes mouse lifespan. Cancers hijack many of the body’s natural pathways so this finding is to be expected.
As a rule of thumb, activation of stress resistance pathways promotes the progression of cancer while reducing the initiation of cancer. The same may be true for taurine which was found to be cytoprotective in other studies. It is important to follow-up these findings and take them seriously; but there is no need to panic.
The future of taurine as a nutraceutical - why I am still excited
As a disclaimer, I started taking taurine after the Singh study and recommended the inclusion of taurine to different supplement companies due to what I perceived as a positive risk/benefit ratio (for example in this one).
First of all, we have to ask, if we strip away all the data from the Singh paper except the mouse lifespan study, does this materially alter the conclusions?
In principle, it is not relevant whether a molecule increases, decreases or stays unchanged with aging if it extends lifespan. The age-related trajectory helps us understand the underlying biology better but it does not affect the result. Any pattern here is compatible with biology. If a molecule declines, supplementation fixed a deficiency. If a lifespan-extending molecule increases with age, then it probably increases as an adaptive response.
My major concern is that the paper suffers from lack of reproducibility on two different fronts (longitudinal biomarker data and invertebrate lifespan data). Does this weaken the data that was not yet called into question? Certainly, to some extent. It is undeniable that we have to update our assessment towards the negative. On the other hand, replication of C. elegans data is notoriously hard so we should not hold this against the authors too much. Not all lifespan-extending interventions will be conserved, either, so a failure in worms is not a showstopper.
Perhaps the authors just got unlucky.
I remain cautiously optimistic about taurine and hope that it will turn out to be a lifespan-extending molecule. My suggestions would be the following:
The authors of the original paper should work on a rebuttal or follow-up.
The ITP should test whether taurine extends the lifespan of HET3 mice. While not able to answer whether the original data cleanly replicates in B6 mice, this will nevertheless address whether the data is robust in mice as a species. And, indeed, taurine has been selected for the most recent round of ITP lifespan studies.
The CITP should also test taurine to test whether T affects lifespan of a wide range of C. elegans strains, and related species, but I do have reservations about the CITP and worms in general. For example, rapamycin works well in our hands in worms while the CITP never got it working. What does it all mean except that worm studies are highly variable? I just don’t know.
Planned human trials with taurine as a geroprotector should go ahead (e.g. TauAge, NCT06613542), because it is a very promising compound, but I would be slightly more hesitant going forward if I were a reviewer.
Why do I still take taurine?
My strategy towards supplementation is two-pronged. On the one hand, I select supplements that have robust mouse lifespan or anti-cancer data. On the other, I aim to take supplements which have good human evidence. Of course, this does not mean human lifespan or mortality outcomes. That would be unrealistic. However, it does mean I am looking for supplements that improve validated cardiometabolic risk factors like blood pressure or lipids. Taurine is one supplement that meets this bar. For example, a recent study from China enrolling 165 type 2 diabetics found that taurine led to a substantial reduction in blood pressure that can rival blood pressure-lowering medications (Li et al. 2025). More broadly, meta-analytic evidence also suggests taurine supplementation can improve several metabolic syndrome–related risk factors.
Acknowledgments
We thank our friends from Xandro lab for supporting the local longevity community in Singapore.
References
Singh, Parminder, et al. "Taurine deficiency as a driver of aging." Science 380.6649 (2023): eabn9257.
Fernandez, Maria Emilia, et al. "Is taurine an aging biomarker?." Science 388.6751 (2025): eadl2116.
Marcangeli, Vincent, et al. "Experimental Evidence Against Taurine Deficiency as a Driver of Aging in Humans." Aging Cell: e70191.
Mayerhofer, Christina, and David T. Scadden. "Niche-generated taurine is leukemic fuel." Cell Research (2025): 1-2.
Sharma, Sonali, et al. "Taurine from tumour niche drives glycolysis to promote leukaemogenesis." Nature (2025): 1-10.
Dawson Jr, Ralph, et al. "The effects of taurine in a rodent model of aging." Taurine 2: Basic and Clinical Aspects. Boston, MA: Springer US, 1996. 37-50.
Eppler, Baerbel, and Ralph Dawson Jr. "The effects of aging on taurine content and biosynthesis in different strains of rats." Taurine 3: Cellular and Regulatory Mechanisms. Boston, MA: Springer US, 1998. 55-61.
Pierno, Sabata, et al. "Chronic administration of taurine to aged rats improves the electrical and contractile properties of skeletal muscle fibers." The Journal of pharmacology and experimental therapeutics 286.3 (1998): 1183-1190.
Li, Yingsha, et al. "Taurine ameliorates blood pressure and vascular function in patients with type 2 diabetes: Randomized, double-blind, placebo-controlled trial." iScience 28.6 (2025).
(1) At least the authors did not include any very young rats, which could exaggerate the results because taurine levels are elevated in human children, as per the discussion in the de Cabo paper.